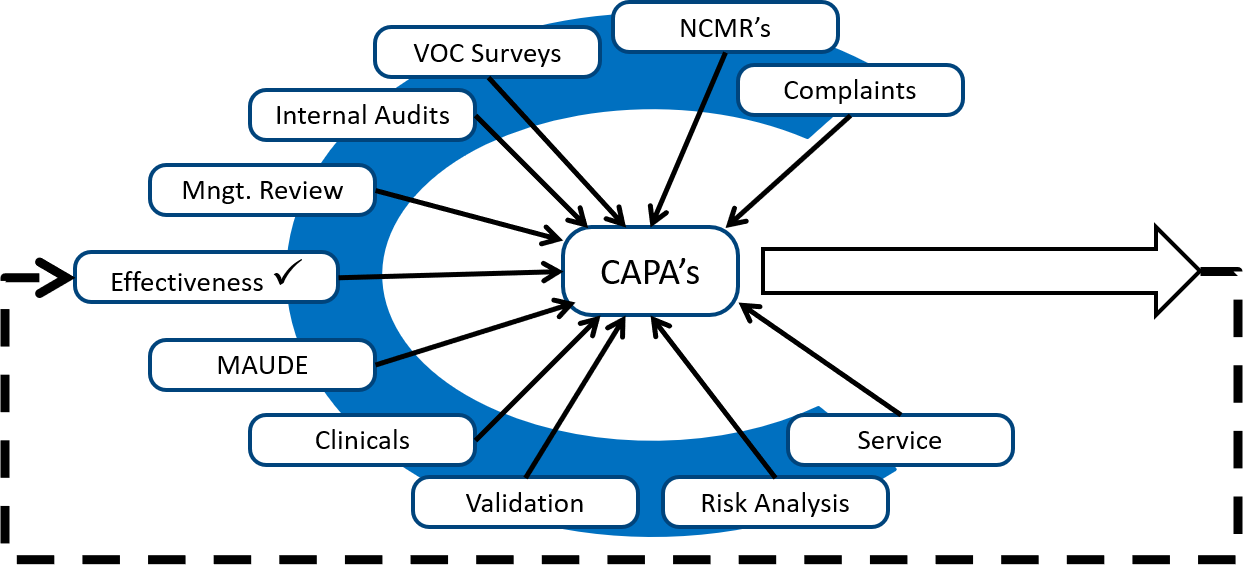
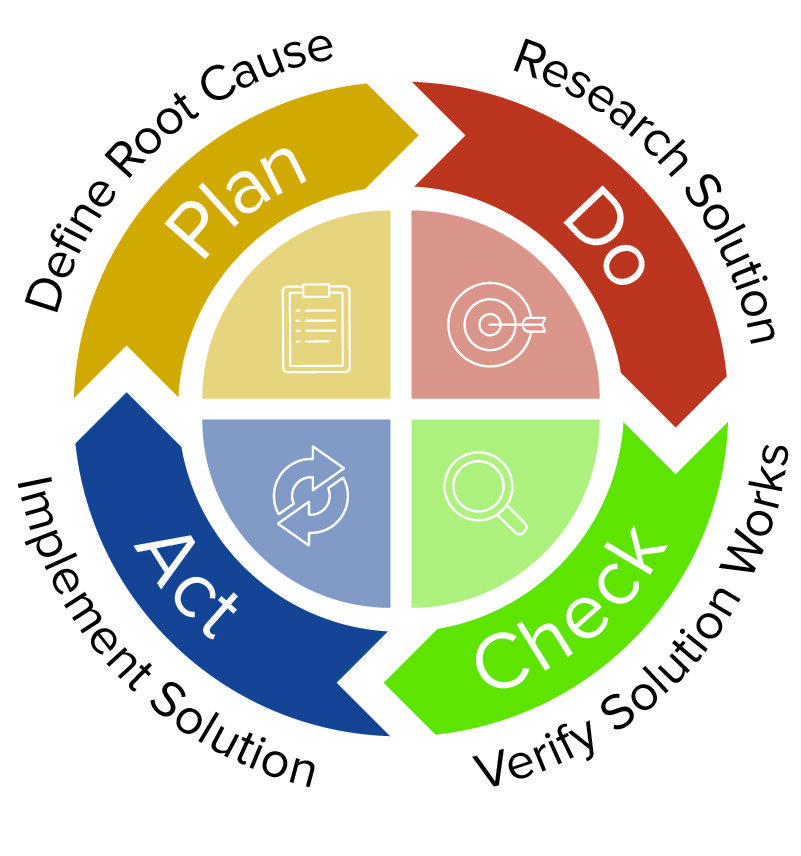
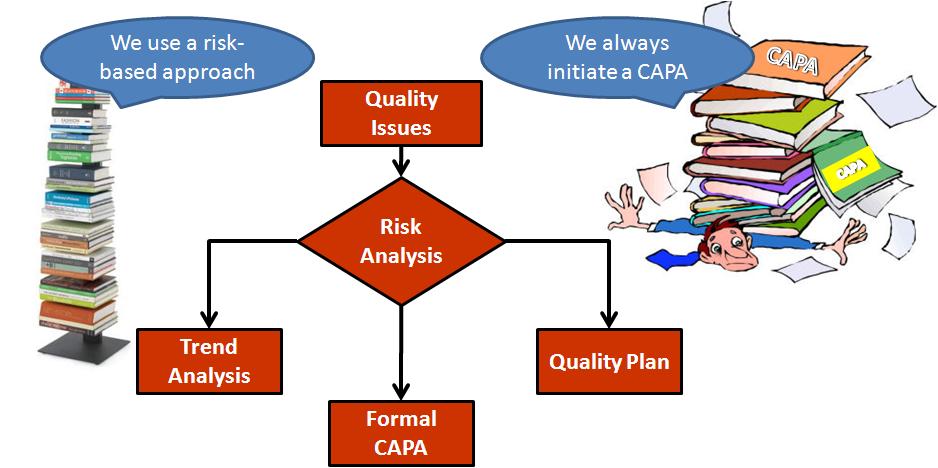
CAPA Certification Assistance | Qualitas Compliance Raleigh | Medical Device Compliance Consulting, ISO 13485, Durham, Chapel Hill, RTP, Boston, Charlotte, Atlanta, Pittsburgh, Tampa, medical device risk management, fda submissions, gap analysis, CAPA

Medical Device CAPA Process : Importance, benefits and implementation | Engineering - Johari Digital Healthcare Ltd.

Medical Device CAPA Process : Importance, benefits and implementation | Engineering - Johari Digital Healthcare Ltd.

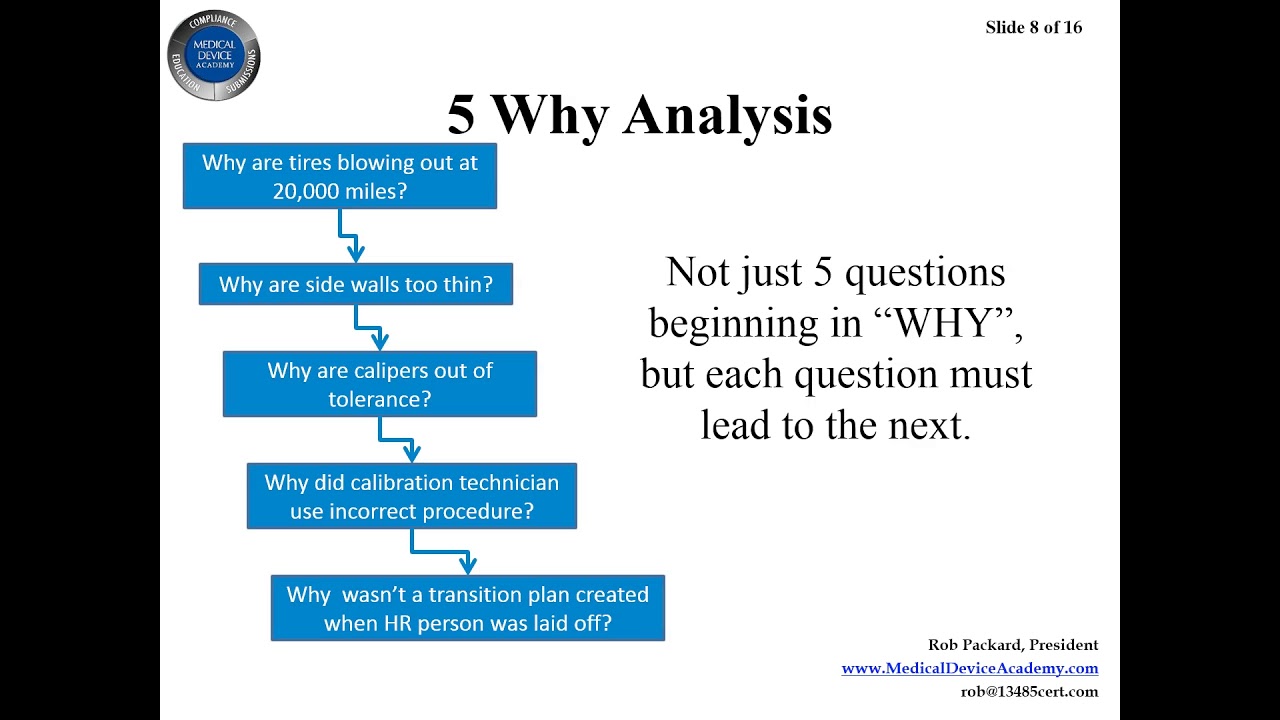
![Corrective and Preventive Action (CAPA): The Definitive Guide [2023] Corrective and Preventive Action (CAPA): The Definitive Guide [2023]](https://www.thefdagroup.com/hs-fs/hubfs/Screen%20Shot%202021-04-26%20at%2012.58.17%20PM.png?width=1229&name=Screen%20Shot%202021-04-26%20at%2012.58.17%20PM.png)