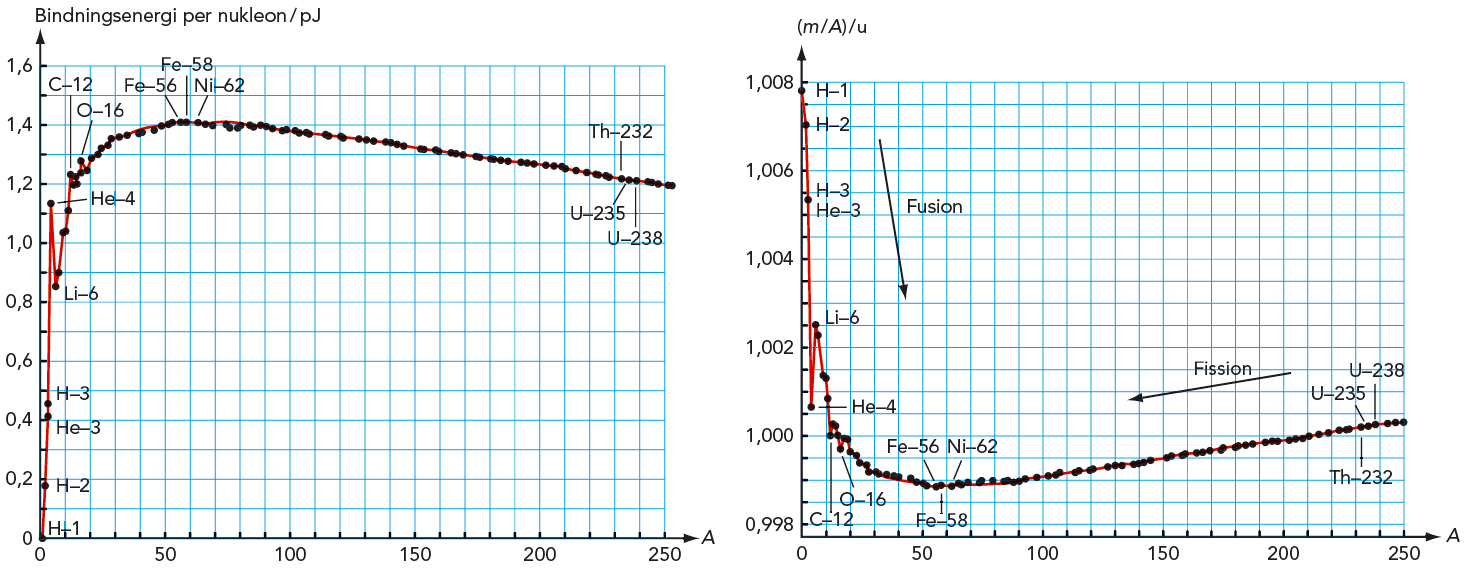
The binding energy of an imaginary iron ^5636Fe is (Given atomic mass of Fe is 55.9349 amu and that of hydrogen is 1.00783 amu. Mass of neutron is 1.00876 amu)
Find BE per nucleon of 56 Fe where m(56 Fe) = 55.936 u mn = 1.00727 u, mp = 1.007274 u - Sarthaks eConnect | Largest Online Education Community